


Dr. Samaneh Shahgaldi is an Associate Professor at the University of Quebec and an Adjunct Associate Professor at the University of Waterloo. She holds the Canada Research Chair (CRC) at proton exchange membrane (PEM) fuel cells and electrolyzers. She is a member of The Institute of Innovations in Ecomaterials, Ecoproducts and Ecoenergy, based on biomass (I2E3) and Hydrogen Research Institute (HRI) of the Université du Québec à Trois-Rivières (UQTR). She is an award-winning researcher, Chair of the scientific committee, and a member of the editorial board of the International Journal of Green Energy. She was also a Senior Research Scientist at Cummins/ Hydrogenics, dealing with different Fuel Cell and Water electrolyzer projects. She is leveraging all her industrial experiences and academic knowledge to improve clean energy technologies.








A proton exchange membrane (PEM) fuel cell consists of a cathode, an anode, and an electrolyte membrane. Hydrogen is oxidized at the anode, and the oxygen is reduced at the cathode. Protons are transported from the anode to the cathode through the electrolyte membrane, and the electrons are carried over an external circuit load.
PEM fuel cells have high energy densities of up to 39.7 kW/kg and relatively low operating temperatures, which makes them easier to contain and reduces thermal losses, smaller volume, and lighter weight making them perfect for automotive and portable applications. These applications include automotive, stationary, portable, and military power generation. We are working on different components to address the three main challenges which are cost, performance, and durability.
Currently, we are working with different industrial partner to develop novel:




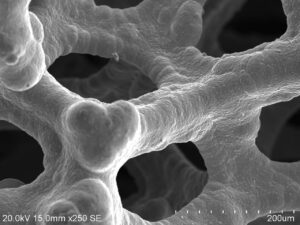

A Proton Exchange Membrane (PEM) water electrolyzer cell is an electrochemical device that uses an external electrical power source to split water into hydrogen and oxygen gases through a process called electrolysis. The electric current is passed through the water, causing the water molecules to split into hydrogen ions (H+) and hydroxide ions (OH-) at the anode and cathode, respectively. The PEM allows only the protons to pass through to the cathode, where they combine with electrons from the external circuit to form hydrogen gas. The hydroxide ions combine at the anode with water to form oxygen gas and more protons.
The electrolyzer cell stack has several components including a cathode, an anode, a selectively permeable proton exchange membrane, as well as cell separators or bipolar plates, and flow distributors such as porous transport layers (PTL’s) and Gas diffusion layer (GDL). PEM electrolysis has many advantages compared with other electrolysis methods; the biggest advantages are the formation of high-purity hydrogen, a small carbon footprint and compact design, the application of higher current density on the electrodes and a high-pressure operation up to 200 bar. Compared to alkaline devices, they are less caustic, can be reversible devices and are able to operate at lower cell voltages, higher current densities, higher temperatures, and pressures leading to higher efficiencies (80–90%).
PEM water electrolysis has been identified as a key process for transforming zero-carbon electricity sources into the supply of zero-carbon hydrogen and oxygen for miscellaneous end uses. In our group, we are focused on improving the performance, durability, and cost-effectiveness of the technology. We are working to develop:





An anion exchange membrane (AEM) water electrolyzer represents a cutting-edge electrochemical device designed to split water into hydrogen and oxygen gases. This innovative technology operates efficiently at room temperature and atmospheric pressure, utilizing non-precious metal electrodes and a solid anion exchange membrane. The membrane’s unique characteristic enables the selective passage of negatively charged ions (anions) while blocking positively charged ions (cations). This feature facilitates the movement of protons from the anode to the cathode while preventing the migration of hydroxide ions (OH-) from the cathode to the anode. Unlike traditional electrolyzers, AEM electrolyzers can operate at higher pH levels, thereby reducing the need for expensive catalyst materials like platinum. Furthermore, they exhibit superior resistance to alkaline conditions, leading to reduced degradation and potentially longer lifetimes, ultimately resulting in more cost-effective systems.
The immense promise of AEM water electrolyzers lies in their ability to produce hydrogen gas as a clean and sustainable energy carrier. This breakthrough technology holds diverse applications in renewable energy storage, fuel cells, and various industries. As a dedicated team, our focus centers on enhancing the efficiency, durability, and cost-effectiveness of different components within the AEM electrolyzer. By relentlessly pursuing advancements in these areas, we aim to pioneer the next generation of water electrolyzers, paving the way for a greener, more sustainable energy landscape and accelerating the transition towards a cleaner and brighter future.




Lignin, a complex organic polymer abundant in plant cell walls and second only to cellulose as the most prevalent natural polymer on Earth, holds immense promise as a renewable feedstock for the production of high-value chemicals and materials. Its applications extend to electrolyzers and fuel cells, where lignin can serve as a sustainable carbon source. In the context of electrolyzers, lignin can undergo conversion into hydrogen-rich feedstocks like syngas or methanol, which then become inputs for the electrolysis process. This enables the production of hydrogen without relying on fossil fuels, fostering a greener and more sustainable hydrogen generation method.
Fuel cells also benefit from the utilization of lignin as a carbon-based catalyst support material. By processing lignin into activated carbon, it becomes an ideal support material for fuel cell catalysts. This application enhances the performance of fuel cells while simultaneously reducing their overall cost, making fuel cell technology more accessible and environmentally friendly. Moreover, lignin finds additional relevance in fuel cell components’ production, including electrodes and membranes. For instance, lignin can serve as a precursor for the creation of carbon nanofibers and act as an electrode material in fuel cells, advancing the development of renewable and efficient energy technologies.
Incorporating lignin into the realm of electrolyzers and fuel cells presents an exciting opportunity to accelerate the progress of sustainable and renewable energy technologies, contributing to a cleaner and more eco-friendly energy landscape.

Lithium-ion batteries serve as rechargeable energy storage devices widely employed in various applications like portable electronics, electric vehicles, and renewable energy systems. These batteries comprise three primary components: the anode (negative electrode), the cathode (positive electrode), and the electrolyte.
The anode in lithium-ion batteries is typically constructed using carbon-based materials, notably graphite, known for their capacity to intercalate lithium ions during charge and discharge cycles, thereby enabling the reversible storage and release of lithium ions. However, to achieve higher energy density, faster charging rates, and enhanced safety, ongoing research is exploring alternative anode materials. Scientists are dedicatedly working on developing materials with a higher theoretical capacity that exhibit minimal volume expansion during lithium-ion insertion and extraction. This research involves investigating various metal alloys and bio-based materials that can potentially meet these objectives.
On the other hand, the state-of-the-art cathode typically consists of transition metal oxides, such as lithium cobalt oxide (LiCoO2), lithium iron phosphate (LiFePO4), or lithium nickel manganese cobalt oxide (LiNiMnCoO2). The selection of the cathode material depends on factors like energy density, cost, and desired performance characteristics. Ongoing efforts in cathode development focus on achieving higher energy density, improved cycling stability, reduced costs, and enhanced safety.
The electrolyte plays a crucial role in facilitating the movement of lithium ions between the anode and cathode during the charge and discharge process. Solid-state electrolytes are garnering attention due to their advantages, including improved safety, higher energy density, and broader operating temperature ranges. These electrolytes can be composed of ceramics, polymers, or composites, and researchers are exploring various material combinations to optimize their ionic conductivity and stability. Additionally, there is active research in developing bio-based materials for energy storage devices, further enhancing the sustainability and potential applications of lithium-ion batteries.

Our laboratory is involved in research primarily focused on energy conversion and storage, valorization of biomaterials, and developing advanced materials for different applications.We are committed to improving clean energy technologies and making them more affordable.